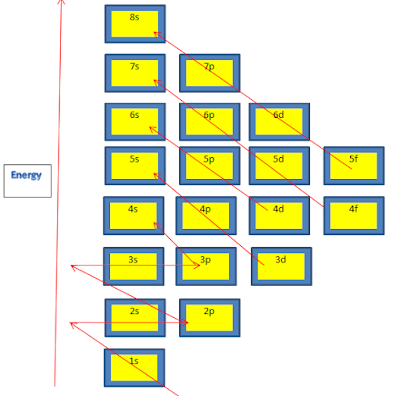
Hydrogen has one electron with almost the same energy level as the nucleus.Let’s take the example of a Hydrogen atom to understand this. When an orbital shell is full, it is represented by the symbol ↑↓. 
Therefore, if one electron is assigned as a spin-up (ms = +1/2) electron, the other electron must be a spin-down (ms = -1/2) electron. The exclusion principle states that every orbit can hold a maximum of two electrons with spin up and spin down.The second principle that we must follow closely aligns with the Pauli Exclusion principle Next, we start assigning these electrons from the lowest-energy orbital shells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
